Quote:
Originally Posted by OrganicBananac
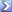
For Richards picture.
1. NOPE
2. I tend to enjoy not paying for someone's repackaging of ingredients, suspect till viewing of ingredient list, then may be proven not guilty.
3. NOPE neon organic,eh?
4. Good stuff, but again there are better less costly alternatives.
|
My
picture was to point that (a) there are chemicals in every fertilizer and (b) it is incorrect that "chemicals kill microbial life".
As for
OrganicBananac's analysis of the picture:
- Inorganic and a poor choice for my soils.
- 100% organic -- you can view the ingredients online with a little effort
- 100% organic -- depending on your definition
- 100% organic -- the cost to consumers varies a lot depending upon location and supplier.
For calculating the true cost of fertilizers, please see this thread:
The Economics of the Fertilizer Numbers
Quote:
Originally Posted by OrganicBananac
... The same goes for nutrient salts, as stated on the wiki quote above because remember the aspect of exactly what those salts are, concentrated. And, "If the medium is hypertonic a concentrated solution, with a lower water concentration than the cell the cell will lose water by osmosis."
|
Nutrient salts such as #3 are never applied in concentrated solution. Dosages range from 100ppm to 1000ppm. At this rate, the nutrients are beneficial to both the plant and soil life.